Featured
Latest News

July 13, 2025
add comment
The Peoples Democratic Party (PDP) has secured a lone councillorship seat in the Lagos State local government...

Peter Obi Deny Defecting to ADC
July 13, 2025
add comment

July 14, 2025
add comment
Popular Nigerian content creator and comedian, Nasboi, has announced his decision to quit skit making after five...

July 14, 2025
add comment
Aisha Buhari, wife of the late former Nigerian President Muhammadu Buhari, has revealed that her husband left...

Cult Violence Claims Another Life in Ogun
July 14, 2025
add comment

July 14, 2025
add comment
Chelsea Football Club will proudly don the World Cup winners badge on their shirt for the next...

July 14, 2025
add comment
Most Asian markets rose on Monday as investors digested Donald Trump’s latest trade war salvos that saw...

June 28, 2025
add comment
The African Export-Import Bank has sealed a partnership deal with the Creative, Reality, Entertainment, Arts and Music...

July 14, 2025
add comment
Dear APC Anambra South Senatorial bye-election delegates, No one goes to the farm with blunt farm tools,...

Visionless APC and Anambra’s Hapless Cockerel
July 11, 2025
add comment
July 12, 2025
add comment
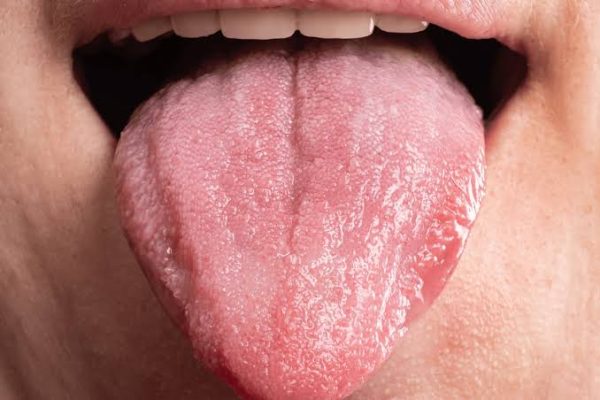
You can notice signs of HIV on the tongue during the early and late stages of the...

Probe Calabar hospital CMD Group Tells Tinubu
July 4, 2025
add comment
July 13, 2025
add comment
The Vice Chancellor of the University of Calabar (UNICAL), Prof. Florence Obi, has directed final-year Dentistry students...